Hepatotoxicity and ADME services



Why us
Our NANOSTACKSTM-based in vitro models recapitulate the cell-cell crosstalk, fluid flow and spatial cellular arrangement typical of the in vivo environment. This way, toxicity screening on our models can yield more accurate results than the use of traditional models. We provide in vitro testing services using the in vitro model of your choice.
Why choose our NANOSTACKSTM liver models
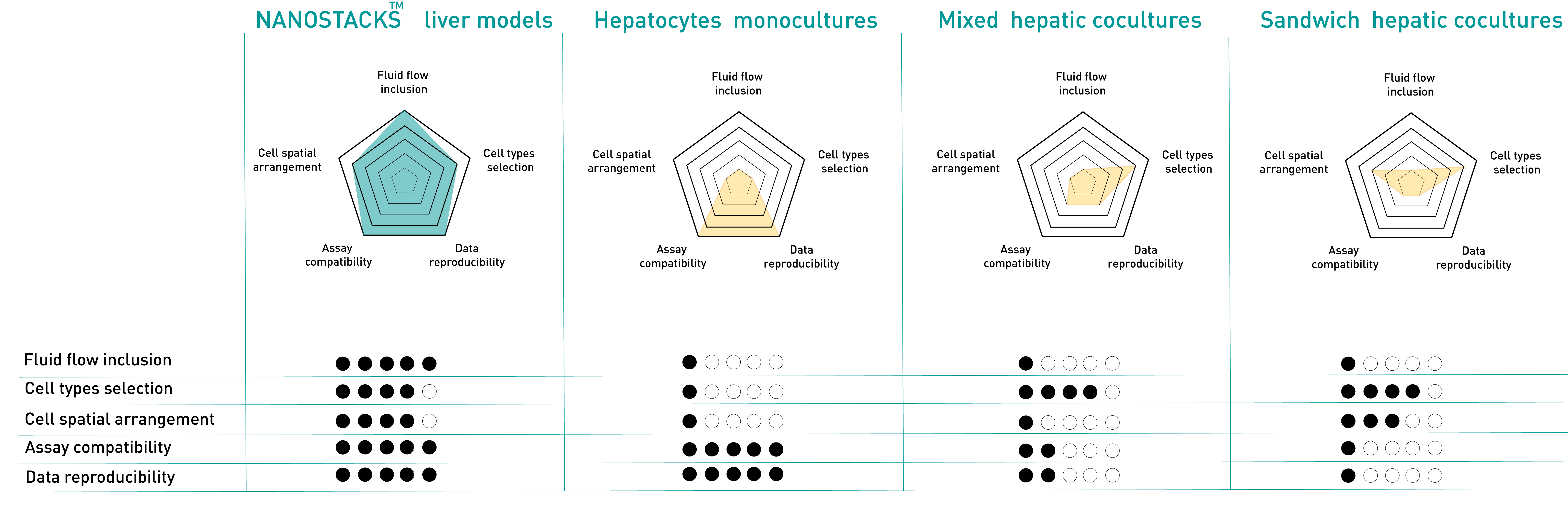
Superior performance compared to other hepatic models
NANOSTACKSTM liver models
NANOSTACKSTM-based models integrate fluid flow, mimicking the liver's mechanical environment, and incorporate various hepatic cell types in a spatially accurate arrangement, including the space of Disse. Unlike standard coculture models where proliferating cells can disrupt reproducibility, NANOSTACKSTM maintain constant cell populations, ensuring consistency. Moreover, isolating cell types on NANOSTACKSTM is straightforward, and they are compatible with assays commonly used for hepatotoxicity and ADME studies.
Hepatocytes monocultures
Hepatocyte monocultures offer consistent results, work well with lab assays and are widely used in pharmaceutical research for tasks like ADME and hepatotoxicity studies. Yet, they lack cell-cell communication with other cell types, in vivo-like spatial organization, and fluid flow typical of the liver environment, limiting their ability to replicate in vivo conditions and the regulation of hepatocyte function.
Mixed hepatic cocultures
Mixed cocultures of hepatocytes and other hepatic cells mimic in vivo cell diversity but lack representative spatial organization. Proliferating cells may dominate unpredictably, challenging reproducibility. Separating cell types for assays is difficult, limiting endpoints. The model lacks fluid flow, hindering replication of the hepatic environment.
Sandwich hepatic cocultures
Sandwich cocultures of hepatocytes and other hepatic cells replicate liver cell diversity and partial spatial distribution seen in vivo. Yet, they lack the space of Disse, featuring unrealistic cell-cell and cell-ECM contacts. Proliferating cells can dominate, reducing reproducibility, and the complex structure complicates isolating cell types for assays. Like other models, they lack fluid flow.
Click on the options below to explore our validated solutions for hepatotoxicity and ADME studies
NANOSTACKSTM liver monoculture model
NANOSTACKSTM liver triculture model
NANOSTACKSTM liver tetraculture model

The model is built using our proprietary NANOSTACKS™ technology
- Layer 1: Hepatocytes compartment
- Cell type: Primary human hepatocytes
- Primary human hepatocytes (PHH) are the gold standard for hepatotoxicity screening due to their physiological relevance and ability to recapitulate key liver functions, such as CYP activity and albumin production. The model can be utilized in various ways, including as a standalone model or as a control for our triculture and tetraculture models.
- Fluid flow included
OUTPUTS
Cytotoxicity
CYP450
Albumin
Urea
Contact us for alternative endpoints.
Comprehensively validated liver monoculture model

Liver PHH cells on NANOSTACKSTM
Representative widefield images of primary human hepatocytes (PHH) in NANOSTACKSTM. Magnification: 10X.

Long term model culture
Primary human hepatocytes (PHH) grown on NANOSTACKSTM under both flow and static conditions maintained viability for 31 days of the study, with no substantial differences between static and flow conditions. Data: mean ± SEM. N = 3 on day 2, 4, 7,11, 14, 20 and 26.
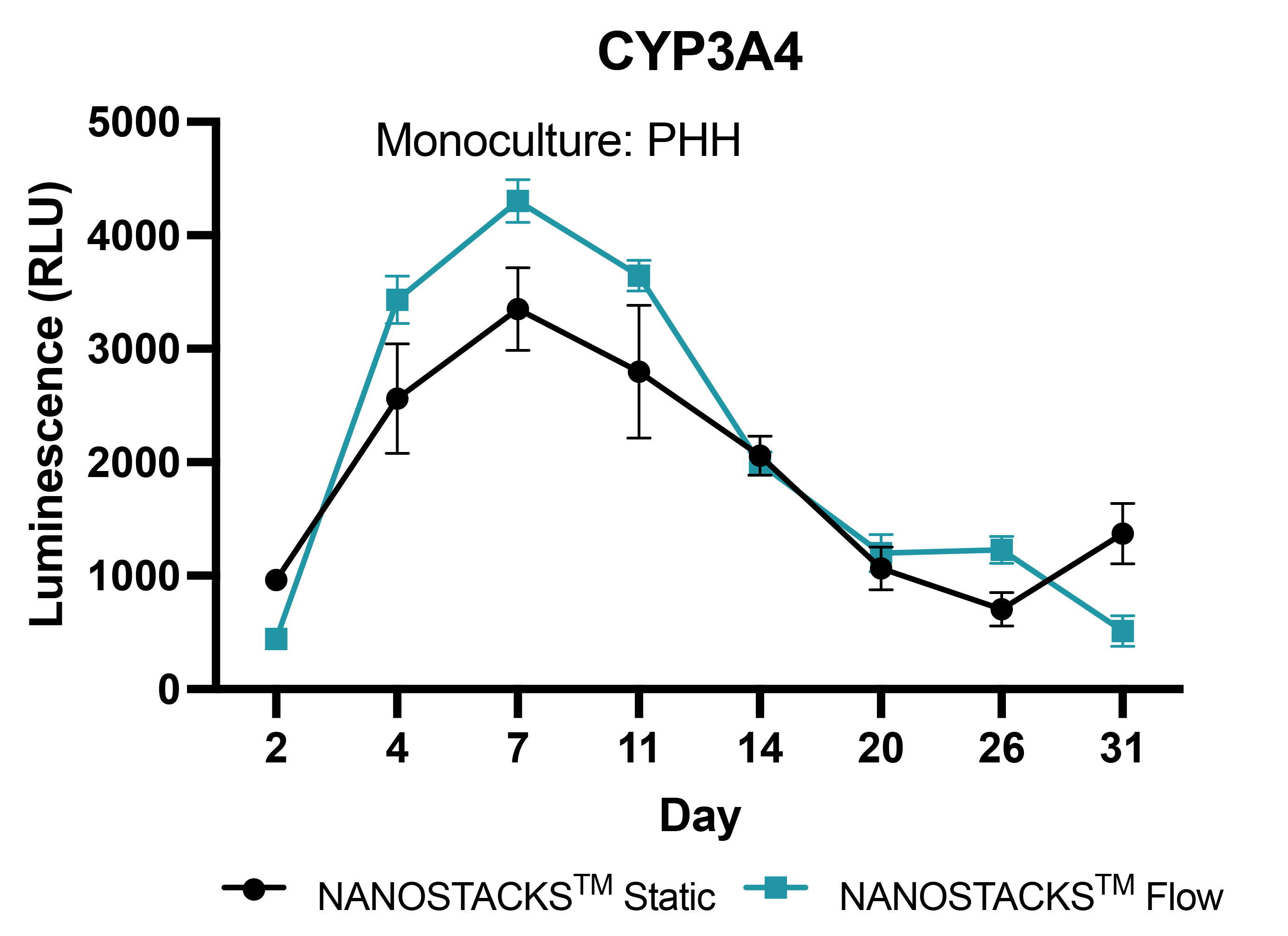
Sustained metabolic competence
The NANOSTACKSTM model of primary hepatocytes (PHH) maintained active metabolic activity throughout the 31-day study period. However, the introduction of flow into the model resulted in significantly higher CYP3A4 activity between days 4 and 14 compared to the static model. Data: mean ± SEM, N = 3 on days 2, 4, 7, 11, 14, 20, 26, and 31.

Human-like albumin levels
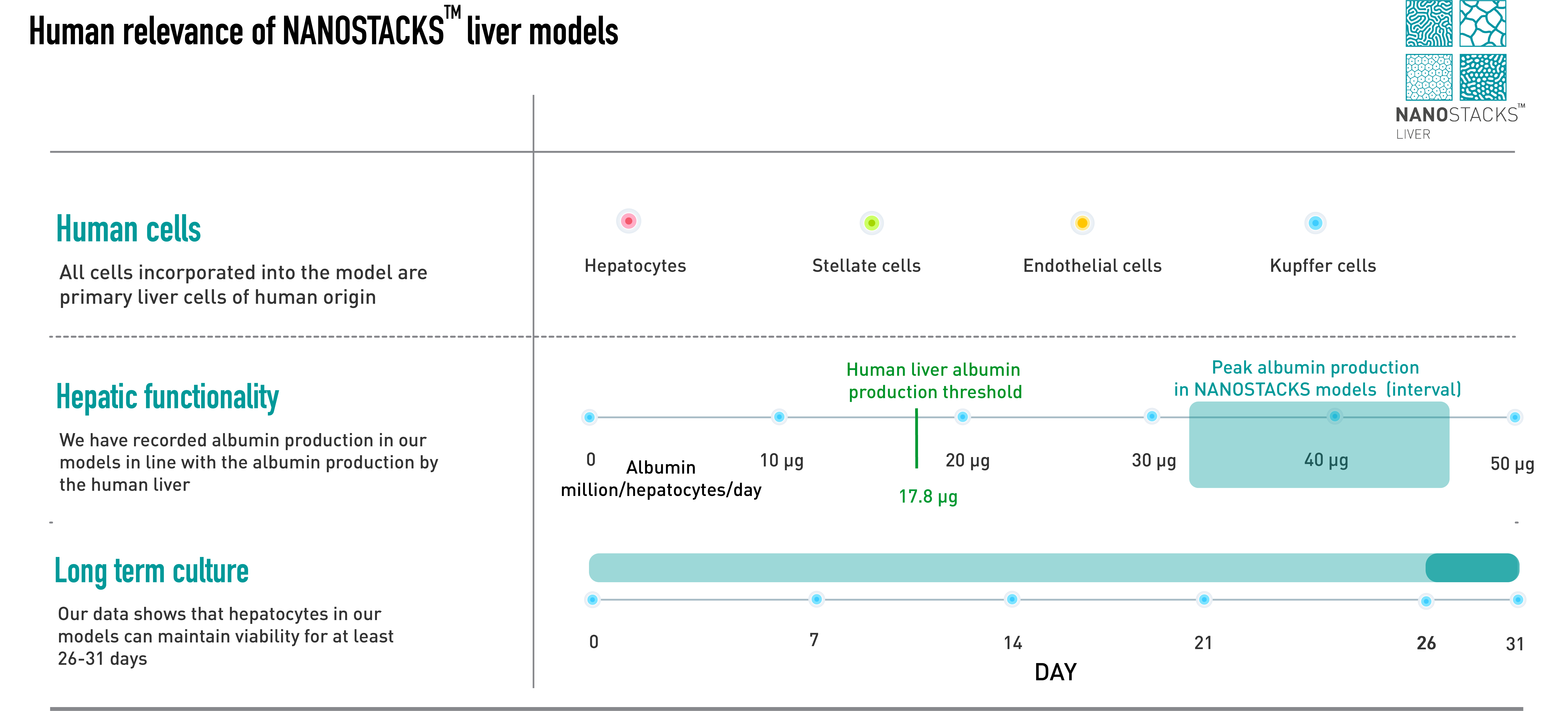
Albumin production in primary human hepatocytes reached human threshold levels (17.8 µg/million/hepatocytes) by day 4 of culture, peaking at approximately 40 µg on day 7. The model maintained above threshold levels until day 26 in both static and flow conditions, except on day 26 when albumin levels in the flow condition decreased to 8.9 µg. Data: mean ± SEM. N = 3 on days 2, 4, 7, 11, 14, 20, and 26.

Human-like urea levels
Under static conditions, human primary hepatocytes (PHH) on NANOSTACKSTM maintained human-relevant levels of urea (>56 µg/million/hepatocytes/day) for 7 days, decreasing to 24.25 µg by day 14. Conversely, when flow was introduced into the model, the human urea level threshold was extended until day 14. Data: mean ± SEM, N = 3 on days 2, 4, 14 and 26.

Clinical translatability
The NANOSTACKSTM human primary hepatocytes (PHH) model accurately predicts hepatotoxicity of Zileuton, an FDA-classified high-DILI-concern drug, at clinically relevant concentrations following 6 days of repeated exposure, demonstrating its robust DILI prediction capability. Data: mean ± SEM. N = 3.
PHH monoculture model DILI screening standard process & timeline*

*Dosing intervals and duration can be adjusted based on your requirements

The model is built on 3 layers of primary liver cells, using our proprietary NANOSTACKS™ technology
- Layer 1: Hepatocytes compartment
- Cell type: Primary human hepatocytes
- Primary human hepatocytes (PHH) are the gold standard for hepatotoxicity screening due to their physiological relevance and ability to recapitulate key liver functions, such as CYP activity and albumin production.
- Layer 2: Stellate compartment
- Cell type: Primary human hepatic stellate cells
- Stellate cells play a major role in liver wound healing, and have a plethora of additional functions, which range from storage of vitamin A to immunity.
- Layer 3: Endothelial compartment
- Cell type: Primary human hepatic endothelial cells
- In addition to regulating the blood flow within the liver, hepatic endothelial cells have a number of important functions, including drug metabolism and detoxification.
- Fluid flow included
OUTPUTS
Cytotoxicity
CYP450
Albumin
Urea
Contact us for alternative endpoints.
Comprehensively validated liver triculture model

Liver cells on NANOSTACKSTM
Representative widefield images of primary human hepatocytes (PHH) in stellate and liver endothelial (LECs)on NANOSTACKSTM layers. Magnification: 10X.
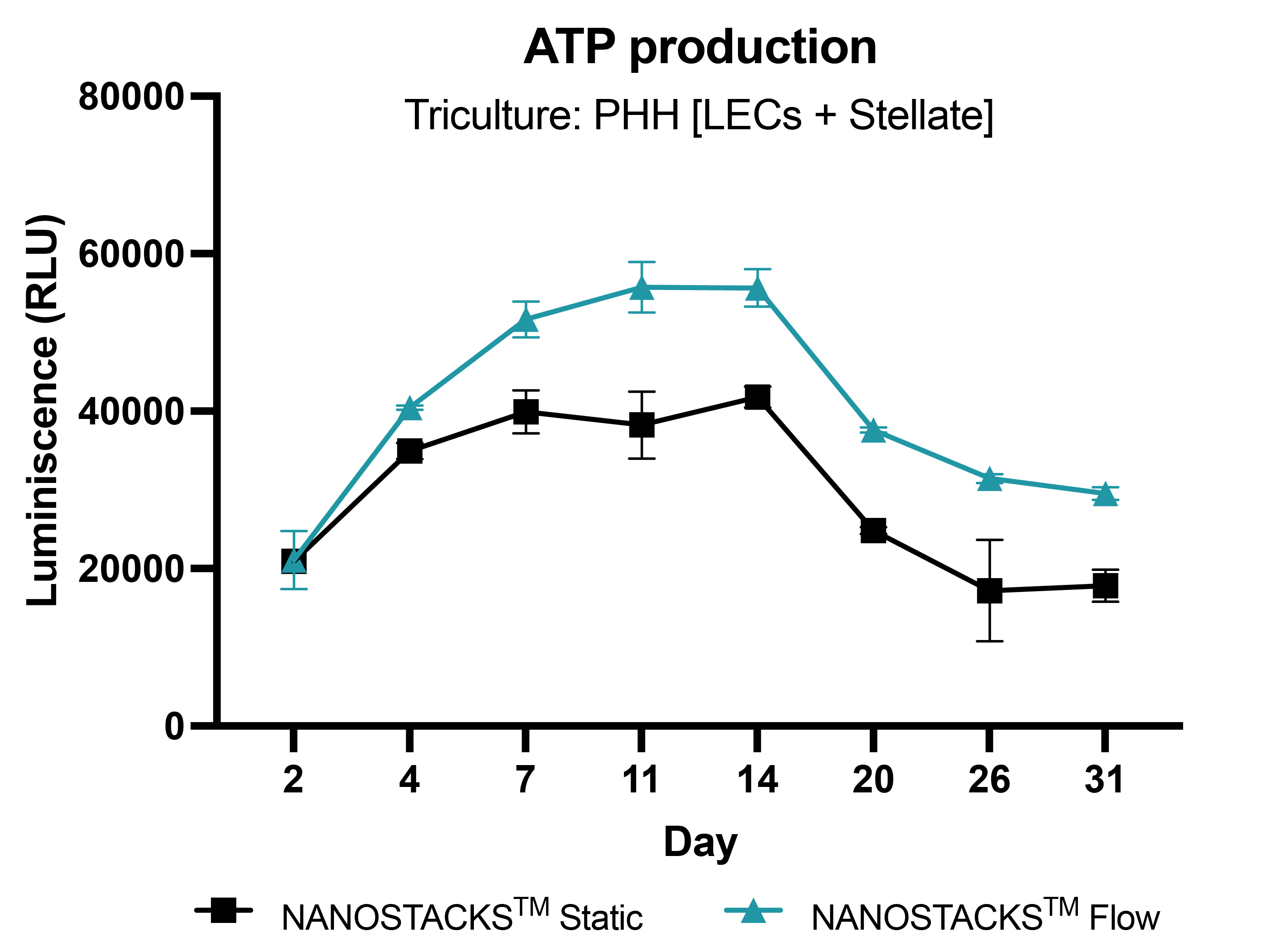
Long term model culture
Primary human hepatocytes (PHH) in triculture with liver endothelial cells (LECs) and stellate cells, grown on NANOSTACKSTM, remained viable for 31 days in the static condition. However, under the flow condition, NANOSTACKSTM exhibited significantly higher viability (ATP production) throughout the entire experiment. Data: mean ± SEM, N = 3 on days 2, 4, 7, 11, 14, 20, and 26.

Sustained metabolic competence
Primary human hepatocytes (PHH) in triculture with liver endothelial cells (LECs) and stellate cells, grown on NANOSTACKSTM, sustained metabolic activity for 31 days in the static condition. However, in the flow condition, CYP3A4 production was up to seven times higher compared to the static condition. Data: mean ± SEM, N = 3 on days 2, 4, 7, 11, 14, 20, 26, and 31.

Human-like albumin levels
Albumin production in primary human hepatocytes (PHH) in triculture with liver endothelial cells (LECs) and stellate cells, grown on NANOSTACKSTM, reached approximate human threshold levels (17.8 µg/million/hepatocytes/day) by day 7 of culture. In the static condition cells generally maintained similar threshold levels for 26 days (around 20 µg). However, albumin production levels doubled in the flow condition, maintaining >43 µg from day 11 to day 26. Data: mean ± SEM, N = 3 on days 2, 4, 7, 11, 14 and 26.
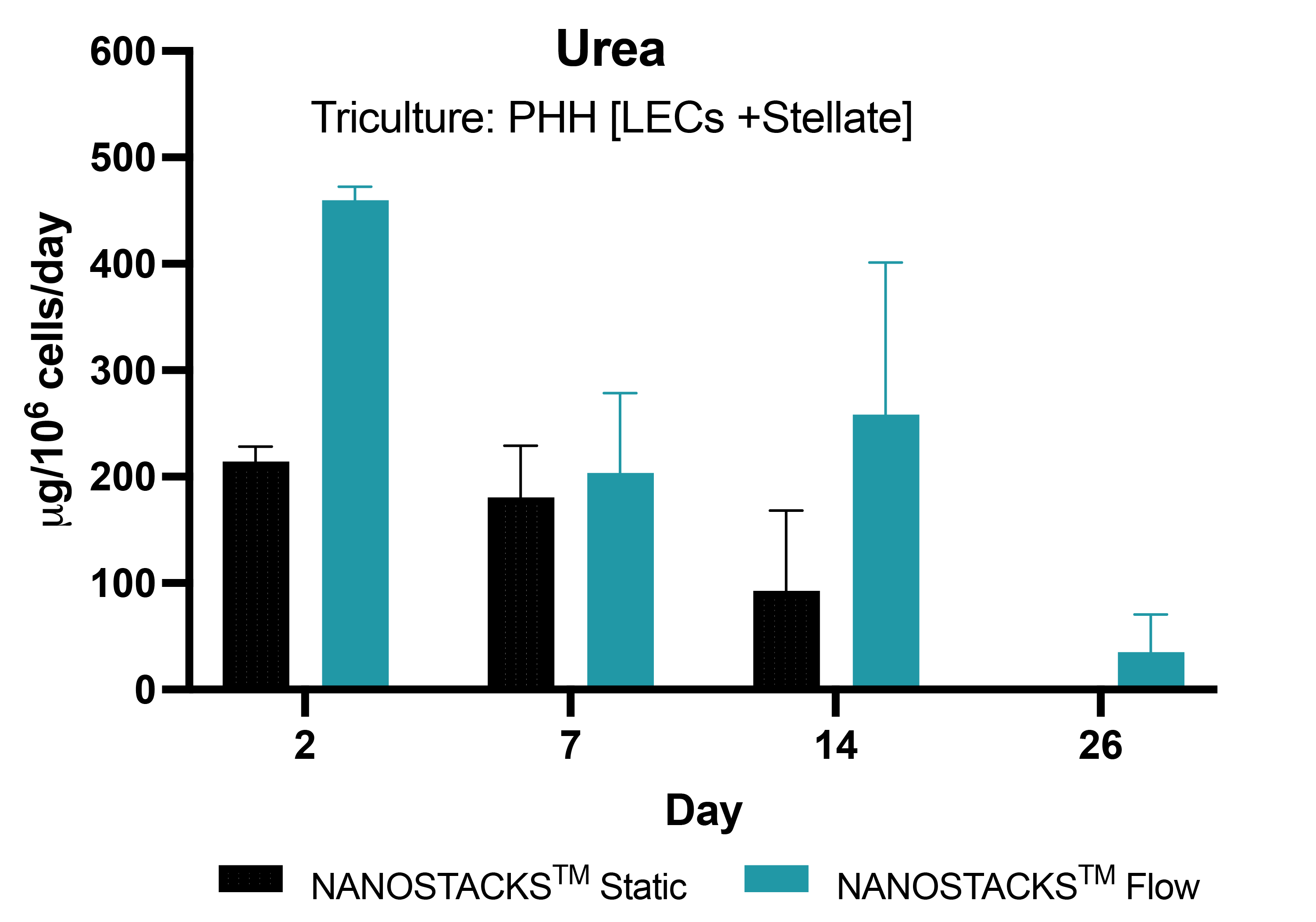
Human-like urea levels
Urea production in primary human hepatocytes (PHH) in triculture with liver endothelial cells (LECs) and stellate cells, grown on NANOSTACKSTM, initially surpassed human threshold levels in early culture, gradually declining over time, and maintained human levels (>56 µg/million/hepatocytes/day) for 14 days under the static condition. However, in the flow condition, urea levels increased up to 2 times and higher levels persisted for 26 days, nearing human urea levels (35.2 µg at day 26). Data: mean ± SEM, N = 3 on days 2, 4, 7, 14 and 26.

Clinical translatability
The NANOSTACKSTM primary human hepatocytes (PHH) in triculture with liver endothelial cells (LECs) and stellate cells, grown on NANOSTACKSTM model accurately predicts the hepatotoxicity of Zileuton, an FDA-classified high-DILI-concern drug, at clinically relevant concentrations following 6 days of repeated exposure (from day 5 to day 10), demonstrating its robust DILI prediction capability. Data are reported as mean ± SEM. N = 3.

Differential toxicity
NANOSTACKSTM allows the examination of the test compounds mechanism of toxicity by assessing the responses on separate individual cell layers. For instance, apart from toxic effects on hepatocytes, Zileuton also induces toxicity in non-parenchymal cells (NPCs) in the model with different dose-responses on stellate cells compared to endothelial cells. Data are reported as mean ± SEM. N = 3.
Triculture hepatic model DILI screening standard process & timeline*
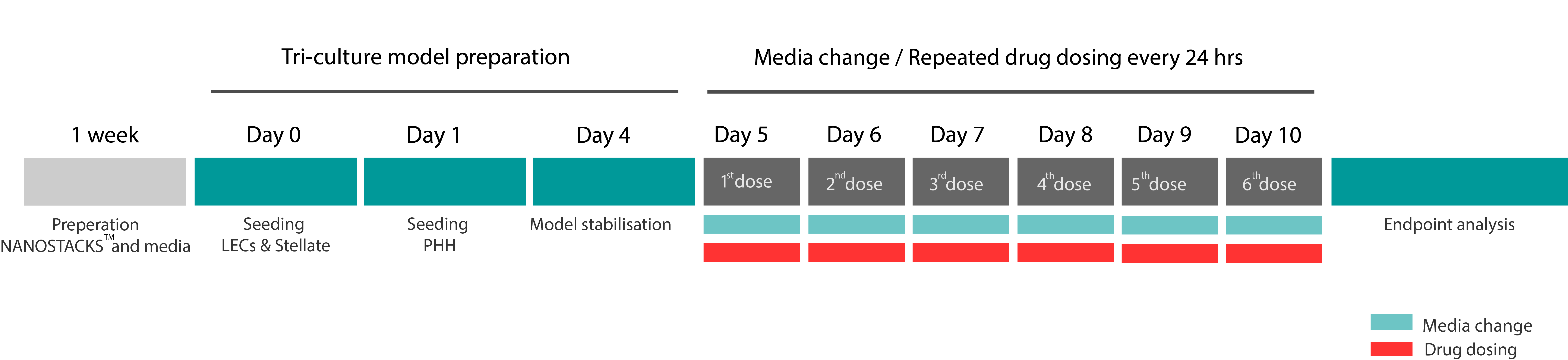
*Dosing intervals and duration can be adjusted based on your requirements.

The model is built on 4 layers of primary liver cells, using our proprietary NANOSTACKS™ technology
- Layer 1: Hepatocytes compartment
- Cell type: Primary human hepatocytes
- Primary human hepatocytes (PHH) are the gold standard for hepatotoxicity screening due to their physiological relevance and ability to recapitulate key liver functions, such as CYP activity and albumin production.
- Layer 2: Stellate compartment
- Cell type: Primary human hepatic stellate cells
- Stellate cells play a major role in liver wound healing, and have a plethora of additional functions, which range from storage of vitamin A to immunity.
- Layer 3: Endothelial compartment
- Cell type: Primary human hepatic endothelial cells
- In addition to regulating the blood flow within the liver, hepatic endothelial cells have a number of important functions, including drug metabolism and detoxification.
- Layer 4: Immune compartment
- Cell type: Primary human hepatic Kuppfer cells
- Kuppfer cells are liver-resident macrophages, and their inclusion in the model is important to replicate aspects of the hepatic immunitary function.
- Fluid flow included
OUTPUTS
Cytotoxicity
CYP450
Albumin
Urea
Contact us for alternative endpoints.
Comprehensively validated liver tetraculture model

Liver cells on NANOSTACKSTM
Representative widefield images of primary human hepatocytes (PHH) stellate, liver enothelial (LECs) and Kuppfer cells on NANOSTACKSTM layers. Magnification: 10X.
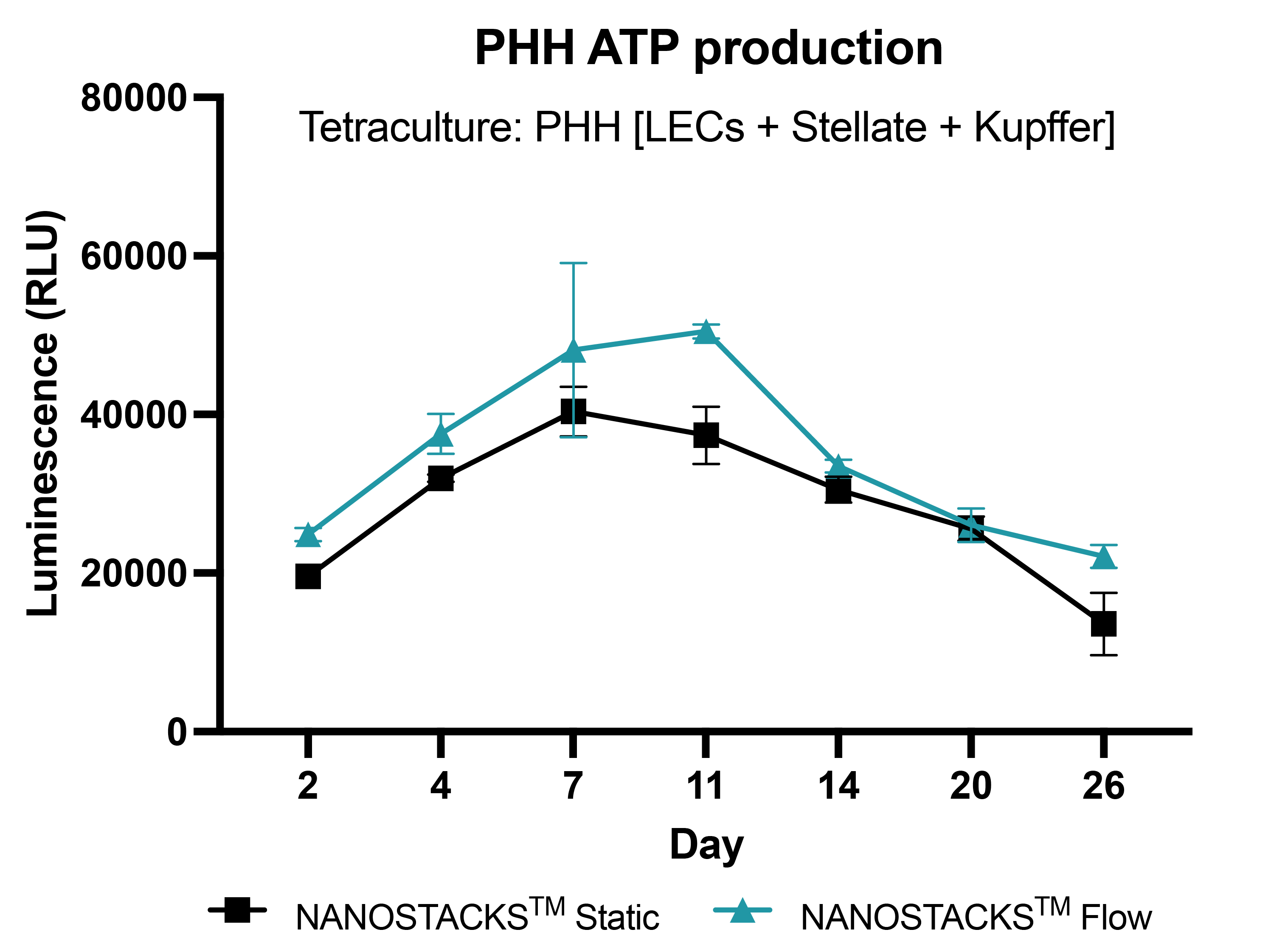
Long term model culture
Primary human hepatocytes (PHH) in tetraculture with liver endothelial cells (LECs), stellate cells, and Kupffer cells, grown on NANOSTACKSTM, maintained viability for 26 days in both flow and static conditions. However, cells under the flow condition generally exhibited higher viability (ATP production) throughout the entire experiment. Data: mean ± SEM. N = 3 on days 2, 4, 7, 11, 14, 20, and 26.
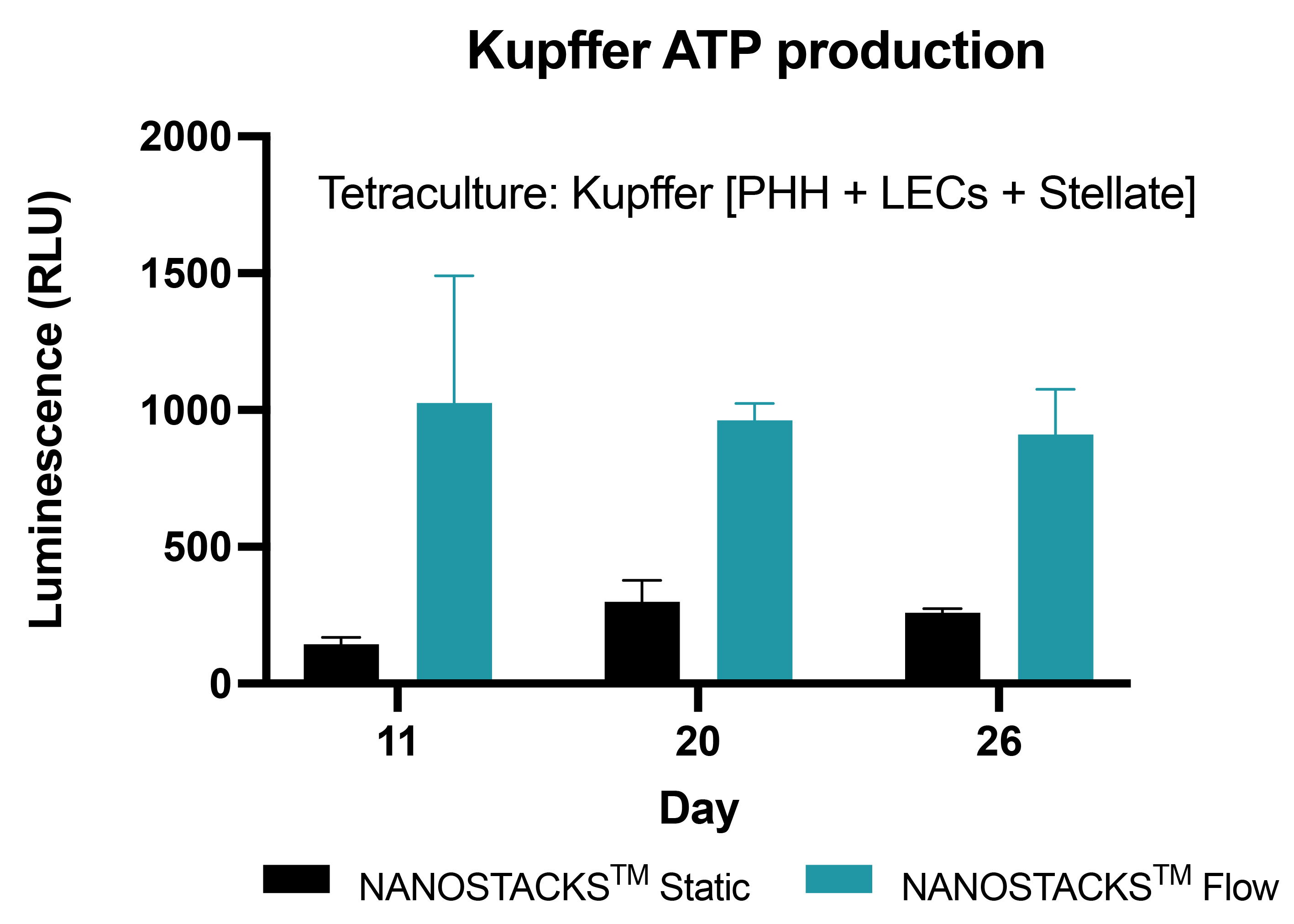
Immunologically active
The immune compartment, including primary human Kupffer cells in tetraculture with human primary hepatocytes (PHH), liver endothelial cells (LECs), and stellate cells on NANOSTACKSTM, maintained viability for at least 26 days. However, introducing flow in the model led to a threefold increase in Kupffer cell viability compared to static conditions. This allows the inclusion of immune interactions with PHH and NPCs in response to drug treatments or in disease modeling. Data: mean ± SEM, N = 3 on days 11, 20 and 26.

Sustained metabolic competence
Primary human hepatocytes (PHH) in tetraculture with liver endothelial cells (LECs), stellate, and Kupffer cells, grown on NANOSTACKSTM, sustained metabolic activity for 26 days under static conditions. However, in the media flow condition CYP3A4 activity was up to five times higher compared to the static condition. Data: mean ± SEM, N = 3 on days 2, 4, 7, 11, 14, 20, and 26.

Human-like albumin levels
Albumin production in Primary human hepatocytes (PHH) in tetraculture with liver endothelial cells (LECs), stellate, and Kupffer cells, grown on NANOSTACKSTM reached approximate human threshold levels (17.8 µg/million/hepatocytes/day) by day 7 and maintained these levels for 26 days of culture in the static condition. A similar trend was noted in the flow condition , with albumin levels reaching higher concentrations on day 11 (33 µg) compared to the static condition. Data: mean ± SEM, N = 3 on days 2, 4, 7, 11, 14 and 26.
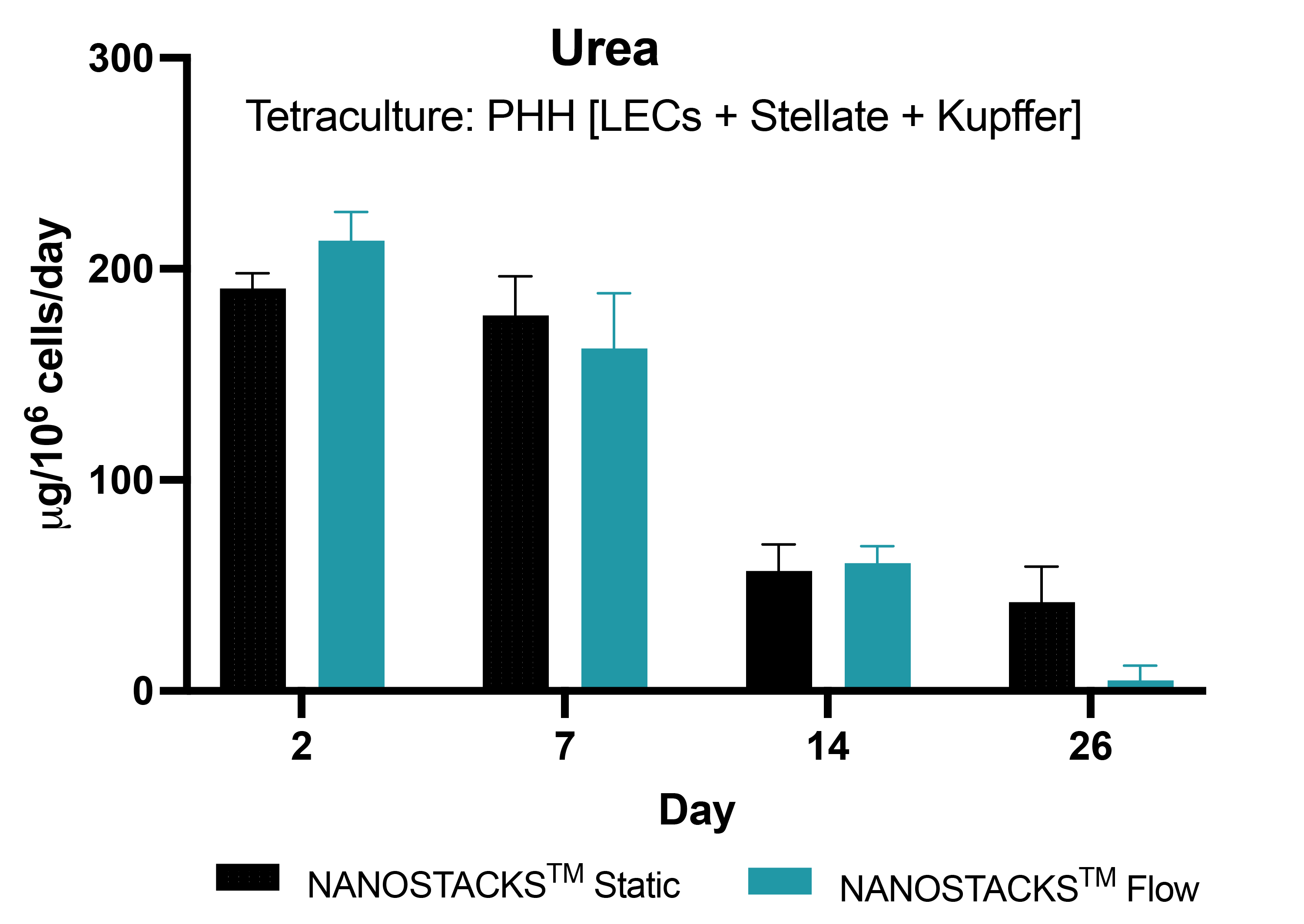
Human-like urea levels
Urea production in Primary human hepatocytes (PHH) in tetraculture with liver endothelial cells (LECs), stellate cells, and Kupffer cells, grown on NANOSTACKSTM peaked in early culture, gradually decreased with time, and consistently stayed above the human level threshold (>56 µg/million/hepatocytes) for 26 days, with similar trends observed in both flow and static conditions. However, by day 26 in the flow condition, urea decreased to 4.9 µg. Data: mean ± SEM, N = 3 on days 2, 7, 14 and 26.
Unique Benefits
Human Relevance
Developed entirely using primary human cells, ensuring translational value with consistent results.

Intercellular communication
Cell-cell crosstalk between key hepatic cells is replicated in the model, increasing its similarity to the in vivo environment.
Inclusion of fluid flow
Fluid flow can be included in the model, modelling the natural mechanical environment of the liver.
Differential toxicity evaluation
Toxicity can be evaluated on each individual cell type included in the model.
Long-term studies
Cells included the model are viable up to 31 days, providing the possibility to perform long-term toxicity screenings.
High quality cells
Our cells are obtained from trusted suppliers and consistently perform key hepatic functions.
Get in Touch
Explore how our models support your research studies

Alternative solutions

MODEL SHIPPING
Alternatively to performing our services within our facilities, we can also ship our validated organ models directly to you lab ready for immediate use.
CUSTOM MODEL DEVELOPMENT
We can develop custom organ models using our NANOSTACKSTM 'Plug&Play' technology based on your requirements.
Need some help?
If you're interested in discovering how our organ models can benefit your research, we invite you to get in touch with us. Our team is available to provide further insights and address any inquiries you may have.
Contact information
UK
Daresbury Laboratory, Sci-Tech Daresbury, Keckwick Lane Daresbury, Cheshire, WA4 4AD
+447588015987
info@revivocell.com
USA
BioLabs@NYU LANGONE, 180 Varick St, Floor 6, New York, NY 10014
info@revivocell.com
Revivocell Brands


